- SAR TECHNOLOGY
- COPPER ISOTOPES
- INTELLECTUAL PROPERTY
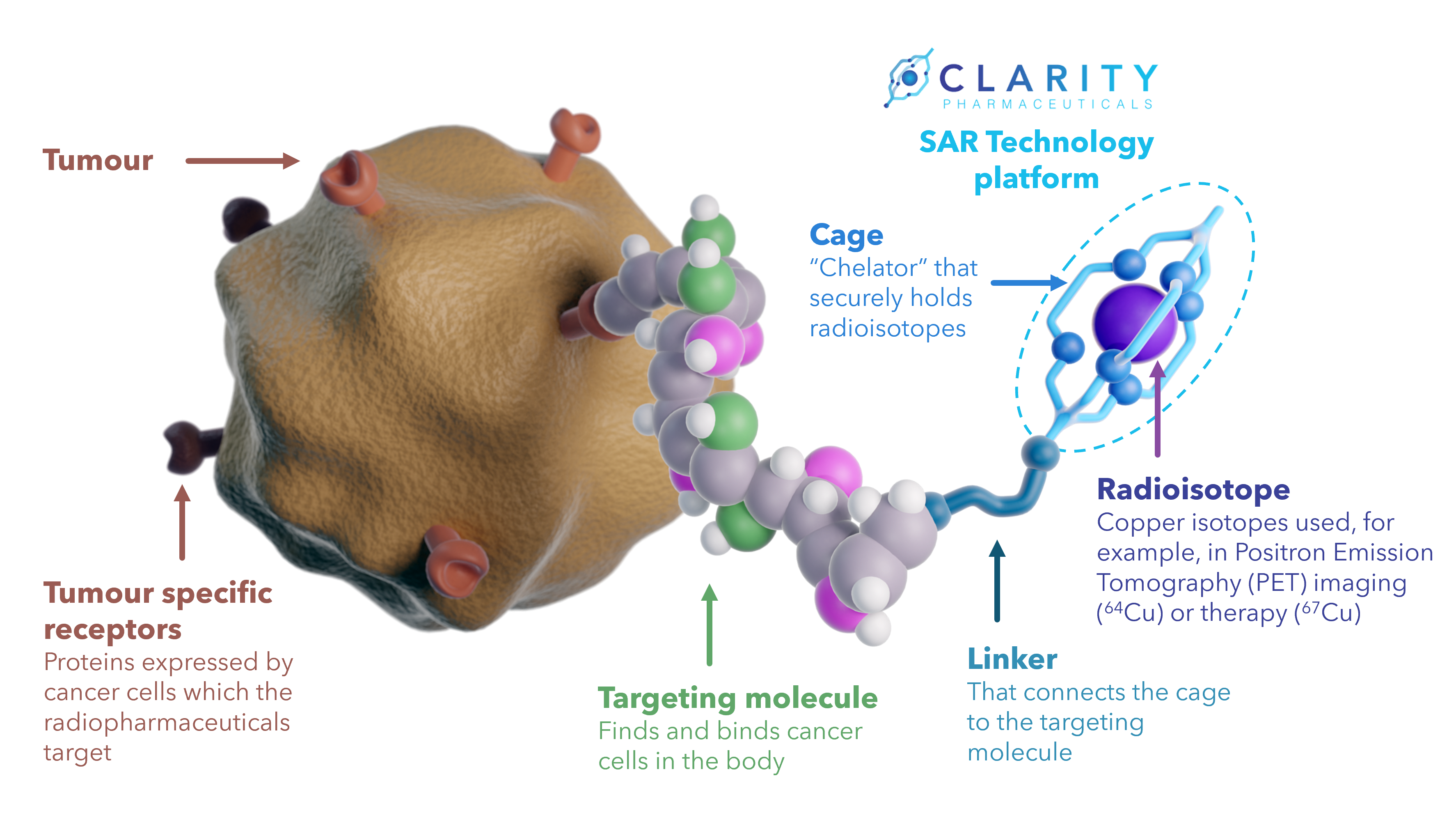
SAR Technology platform
Clarity’s proprietary SAR Technology can be used to develop a range of theranostic radiopharmaceuticals that target different types of cancer. The technology can be employed for diagnostic imaging using (PET) cameras as well as for targeted cancer therapy.
At the heart of Clarity’s theranostic SAR Technology platform is a highly specific and highly stable bifunctional cage (chelator) that retains copper isotopes within it and prevents their leakage into the body. The cage is linked to a targeting molecule, which finds and binds tumour specific receptors on cancer cells. Once the targeting molecule has found the tumour, the radioisotope can act in that location, either emitting radiation to be picked up by an imaging device (diagnostic radioisotope) or by destroying the cancer cells (therapeutic radioisotope).
Together with the targeting molecule and the isotope, the technology enables the development of radiopharmaceuticals for diagnosis and therapy in oncology.
High accuracy and high precision
The two key clinical benefits of using copper isotopes are that they provide high accuracy and high precision in diagnosing and treating cancer.
High accuracy is achieved by only selecting those patients for therapy who show product uptake in the tumours when imaging with a PET scan during the diagnostic stage, an indicator that the product targets the cancer and not healthy tissues, thereby maximising the probability of treatment success.
High precision is achieved by using the same targeting molecule with the same chemical element inside the chelator (a different isotope of copper), ensuring both the diagnostic and therapeutic products have identical targeting in vivo.
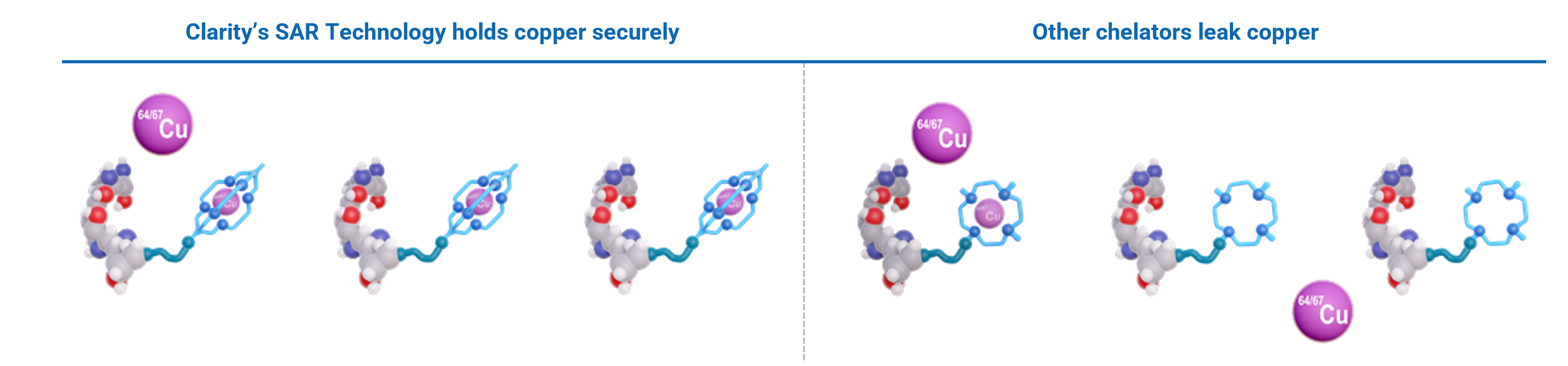
Superior copper cage
The “perfect pairing” of copper-64 (64Cu) and copper-67 (67Cu) isotopes for diagnosis and therapy has been discussed in scientific literature for approximately 25 years. The pairing is well suited for theranostics due to the unique physical characteristics of each isotope of copper as well as having the “ideal theranostic pair”, meaning the same product and same element are used for both diagnosis and therapy.
Historically, the utilisation of copper radioisotopes has been hampered as there were no suitable cages (chelators) that would hold the isotopes inside securely. Earlier attempts with other cages have resulted in the isotopes of copper being released from the cage once injected into the body and then metabolised in the liver, limiting their diagnostic potential and being unsuitable for therapeutic applications.
Clarity seeks to address this issue with its sarcophagine chelator and holds a large IP portfolio on the SAR Technology platform, enabling its commercial exploitation, whereas most other chelators remain unpatented.
Comparison of 64Cu SARTATE and 64Cu DOTATATE
Clarity’s 64Cu SARTATE
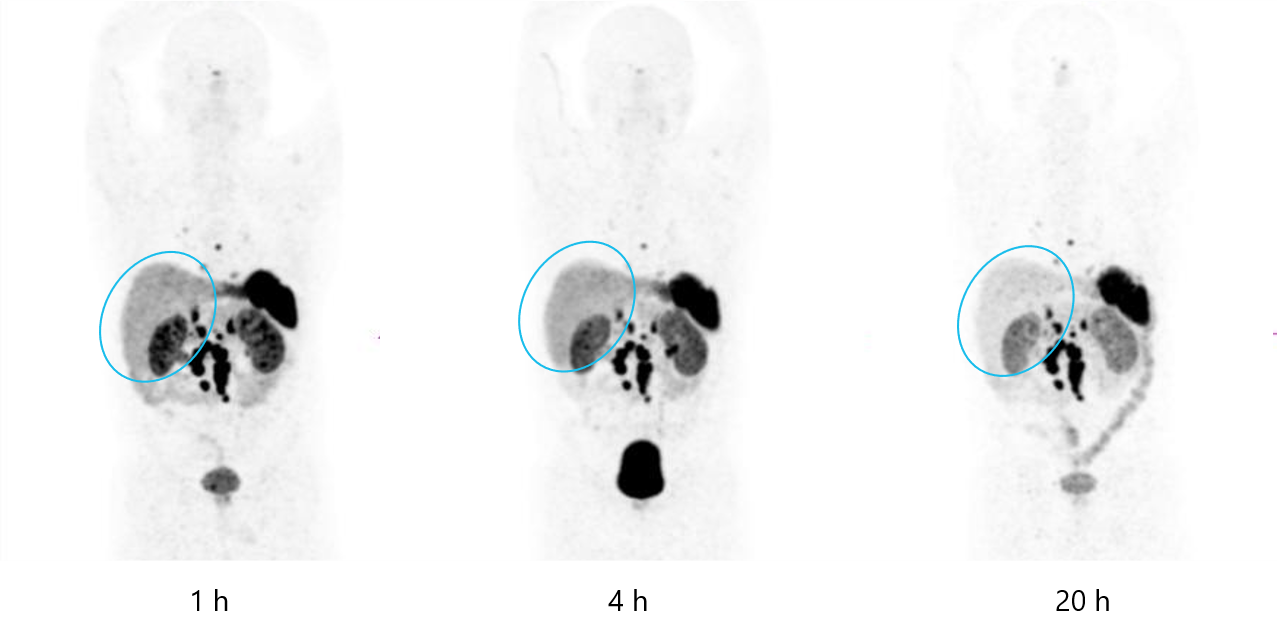
Hicks et al. J Nucl Med 2019; 60:777-785
64Cu DOTATATE
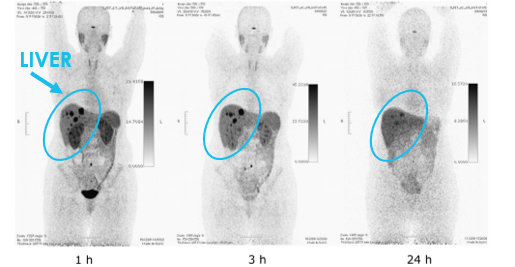
Pfeifer et al. J Nucl Med 2012; 53:1207-15
These images highlight the improvement in clinical outcomes that Clarity’s SAR Technology brings. Free 64Cu leaked from the chelators usually accumulates in the liver, presenting background radioactivity on diagnostic scans. The images of 64Cu SARTATE™ (image on the left) highlight that there is a decrease in detected radioactivity in the liver over time, which is indicative that there is minimal free 64Cu in the body as a result of chelator leakage, meaning that there is minimal copper leaking from the product. In comparison, images with 64Cu DOTATATE (image on the right), which employs a chelator called DOTA, show that there is a constant background in the liver, indicative of the presence of free 64Cu in the liver, meaning there is leakage from the DOTA chelator.
As a result, Clarity’s chelator technology allows PET images to be collected over a wide range of timepoints and, importantly, permits the use of 67Cu-based products for therapy. Utilising the patented SAR technology, Clarity is running a number of clinical trials in a range of cancer types.
Details of our current program can be found in the Pipeline section of the website.
Discovery Program
Clarity’s SAR Technology platform can be used in conjunction with any number of cancer targeting molecules to create new radiopharmaceutical products, which are developed in Clarity’s discovery program.
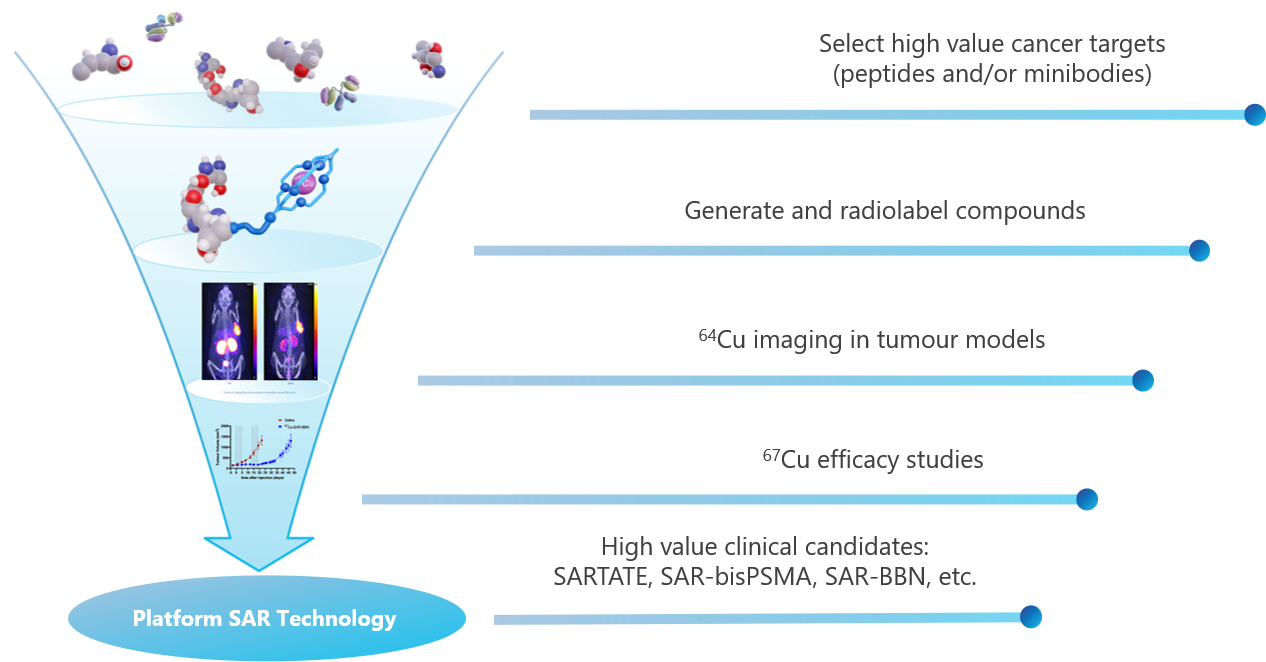
Clarity has a number of research and development projects in its Discovery Program, including:
- New pipeline products; and
- Second generation technology.
The majority of the Discovery work is done in collaboration with Clarity’s academic partners. The funding of the Discovery research is partially supported by grants and the Australian R&D Tax Incentive.
